Atomic probe of bond dynamics and reactivity of individual HF and H2O atoms
August 20, 2020 - A new study of scientists from the Universities of Ulm (Germany), Nottingham (UK), Southampton (UK) and Leeds (UK) enables bond dynamics and reactivity of H2O and HF to be studied at the single-molecule level. The atomic scale study was conducted at low and very low primary energies of 30 - 80 keV in the SALVE microscope and at 60 keV in the SuperSTEM microscope by entrapping HF and H2O molecules within fullerene C60 cages in nanotubes.
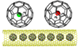
The carbon nanotube is one of the most robust and effective materials for molecular encapsulation in TEM studies. It can restrict molecular movement and can be used as a tiny test tube [1, 2]. When Fullerenes C60, with a cavity similar to a very small nanotube, are entrapped in bigger nanotubes, (Figure 1), they provide an ideal platform for the entrapment of even isolated atoms.[1 - 5]. A team of scientists has now trapped small volatile molecules like H2, HF, and H2O inside C60 in CNT to prevent their rapid molecular movement and to be able to study them at the single molecule level in the TEM. The research helps scientists to better understand the properties of di- or triatomic molecules consisting of light elements. The TEM can also be used to trigger bond dissociation in individual molecules in order to be able to examine chemical bonds and processes at the atomic level.[6, 7] With this approach, the energetics and dynamics of several heteroatomic bonds can be investigated, including CH, CD, CS, CC, Mo-I, and Re-Re at the single-molecule level.[6 - 9]
With the new TEM imaging of the materials, the scientists used 30 keV electron beams (HF @ C60) @ SWNT and (H2O @ C60) @ SWNT. The ordered chains of fullerene molecules appear as circles within the cylindrical channel of the nanotube very similar to the empty C60 in nanotubes. In contrast to the empty C60 fullerenes, which show no observable damage even at higher electron beam energies such as 40 keV, the endohedral fullerene molecules are noticeably crosslinked and polymerized under the irradiation (Figure 2). However, the crosslinking occurs slowly so that useful insight into the mechanisms by which the electron beam is damaged can be studied. With increasing energy of the electron beam from 30 to 80 keV, the stability of the endohedral fullerenes decreases significantly by a factor of ~ 10 for HF @ C60.
Energy Transfer and Bond Dissociation under the Electron Beam
The research also provides an important insight into the state of the caged elements. Figure 3 shows individual F atoms in two intact C60 molecules, while no F signal was detected in polymerized molecules. The diatomic HF and triatomic H2O molecules are enclosed in HF @ C60 and H2O @ C60, respectively, so that the F atom and the O atom are positioned in the center of the icosahedral C60 cage, and retains all three degrees of freedom of rotation. The translational freedom is limited by the fullerene cage to ~ 0.2 nm. [3 - 5] Therefore, the enclosed molecules, (HF @ C60) @ SWNT and (H2O @ C60) @ SWNT, can be described as a free rotator in its original state. A recent theoretical study investigating a hypothetical (HF @ C60) @ SWNT structure concluded that HF molecules in the neighboring HF @ C60 are likely to interact by dipole-dipole coupling on the order of only a few millielectron volts. Overall, the state of the trapped molecules in (HF @ C60) @ SWNT and (H2O @ C60) @ SWNT is closer to the gas state in terms of rotation and vibration, but closer to a solid crystalline state in terms of translation, so that the lateral positions of individual molecules are severely restricted during TEM imaging. The calculated geometry based on the TEM data of HF @ C60 and H2O @ C60 indicates that the geometry of the entrapped small molecules is almost identical to that of the gas phase.
The research also provides an important data point for theoretical modeling of Bond Dissociation and atom permeation through C60. After dissociation from the molecule, the kinetic energy of the hydrogen atom is sufficient to continue its movement across the C60 cavity and through the fullerene wall and prevent recombination with HF @ C60 (Video 7). At lower transmitted energies, the penetration barrier depends on the alignment of the H atom momentum with respect to the carbon-carbon bonds of C60, while dissociation with kinetic energies that are insufficient to emit the hydrogen atom from the fullerene cage reforms the original HF or H2O structure. These results are in agreement with the energy barrier for a hydrogen atom to penetrate through the fullerene wall in the region of 2.6 eV through a hexagon of C60.[10] Therefore, both HF and H2O are effectively depleted of hydrogen atoms under TEM imaging conditions.
The scientists used the SALVE microscope, which offers the capability of performing atomically resolved HRTEM at very low primary energies down to 20 kV as well as the Nion UltraSTEM microscope at the SuperSTEM lab, which offers the capability of performing atomically resolved EELS analysis at 60 kV. They combined the data with DFT simulations to get insight into the fundamental mechanisms of bond dissociation and reactivity of HF and H2O.
"The energy barrier for the O atom penetrating through C60 via an epoxide intermediate was found much lower than the barrier for penetration through graphene. These data offer another important building block for the investigation of the permeability of carbon materials," said Ute Kaiser, head of the SALVE microscopy center.
Resource: Biskupek, J., Skowron, S. T., Stoppiello, C. T., Rance, G. A., Alom, S., Fung, K. L., Whitby, R. J., Levitt, M. H., Ramasse, Q. M., Kaiser, U. A., Besley, E. & Khlobystov, A. N. (2020) Bond Dissociation and Reactivity of HF and H2O in a Nano Test Tube. ACS Nano, 14, 11178, doi: 10.1021/acsnano.0c02661#.
-
Ran, K.; Zuo, J.-M.; Chen, Q.; Shi, Z. Electron Beam Stimulated Molecular Motions. ACS Nano 2011, 5, 3367– 3372, DOI: 10.1021/nn2006909
-
Zhao, J.; Huang, J.-Q.; Wei, F.; Zhu, J. Mass Transportation Mechanism in Electric-Biased Carbon Nanotubes. Nano Lett. 2010, 10, 4309– 4315, DOI: 10.1021/nl1008713
-
Beduz, C.; Carravetta, M.; Chen, J. Y.-C.; Concistre, M.; Denning, M.; Frunzi, M.; Horsewill, A. J.; Johannessen, O. G.; Lawler, R.; Lei, X.; Levitt, M. H.; Li, Y.; Mamone, S.; Murata, Y.; Nagel, U.; Nishida, T.; Ollivier, J.; Rols, S.; Room, T.; Sarkar, R.Quantum Rotation of Ortho and Para-Water Encapsulated in a Fullerene Cage. Proc. Natl. Acad. Sci. U. S. A. 2012, 109, 12894– 12898, DOI: 10.1073/pnas.1210790109
-
Kurotobi, K.; Murata, Y. A Single Molecule of Water Encapsulated in Fullerene C60. Science 2011, 333, 613– 616, DOI: 10.1126/science.1206376
-
Levitt, M. H.; Horsewill, A. J. Nanolaboratories: Physics and Chemistry of Small-Molecule Endofullerenes. Philos. Trans. R. Soc., A 2013, 371, 20130124, DOI: 10.1098/rsta.2013.0124
-
Skowron, S. T.; Chamberlain, T. W.; Biskupek, J.; Kaiser, U.; Besley, E.; Khlobystov, A. N. Chemical Reactions of Molecules Promoted and Simultaneously Imaged by the Electron Beam in Transmission Electron Microscopy. Acc. Chem. Res. 2017, 50, 1797– 1807, DOI: 10.1021/acs.accounts.7b00078
-
Markevich, A.; Kurasch, S.; Lehtinen, O.; Reimer, O.; Feng, X.; Müllen, K.; Turchanin, A.; Khlobystov, A. N.; Kaiser, U.; Besley, E. Electron Beam Controlled Covalent Attachment of Small Organic Molecules to Graphene. Nanoscale 2016, 8, 2711– 2719, DOI: 10.1039/C5NR07539D
-
Chamberlain, T. W.; Biskupek, J.; Skowron, S. T.; Bayliss, P. A.; Bichoutskaia, E.; Kaiser, U.; Khlobystov, A. N. Isotope Substitution Extends the Lifetime of Organic Molecules in Transmission Electron Microscopy. Small 2015, 11, 622– 629, DOI: 10.1002/smll.201402081
-
Botos, A.; Biskupek, J.; Chamberlain, T. W.; Rance, G. A.; Stoppiello, C. T.; Sloan, J.; Liu, Z.; Suenaga, K.; Kaiser, U.; Khlobystov, A. N. Carbon Nanotubes as Electrically Active Nanoreactors for Multi-Step Inorganic Synthesis: Sequential Transformations of Molecules to Nanoclusters and Nanoclusters to Nanoribbons. J. Am. Chem. Soc. 2016, 138, 8175– 8183, DOI: 10.1021/jacs.6b03633
-
Vehviläinen, T. T.; Ganchenkova, M. G.; Oikkonen, L. E.; Nieminen, R. M. Hydrogen Interaction with Fullerenes: From C20 to Graphene. Phys. Rev. B: Condens. Matter Mater. Phys. 2011, 84, 085447 DOI: 10.1103/PhysRevB.84.085447
-
Linck, M.; Hartel, P.; Uhlemann, S.; Kahl, F.; Müller, H.; Zach, J.; Haider, M.; Niestadt, M.; Bischoff, M.; Biskupek, J.; Lee, Z.; Lehnert, T.; Börrnert, F.; Rose, H.; Kaiser, U. Chromatic Aberration Correction for Atomic Resolution TEM Imaging from 20 to 80 KV. Phys. Rev. Lett. 2016, 117, 076101 DOI: 10.1103/PhysRevLett.117.076101
-
Fung, K. L. Y.; Fay, M. W.; Collins, S. M.; Kepaptsoglou, D. M.; Skowron, S. T.; Ramasse, Q. M.; Khlobystov, A. N. Accurate EELS Background Subtraction – An Adaptable Method in MATLAB. Ultramicroscopy 2020, 217, 113052, DOI: 10.1016/j.ultramic.2020.113052
-
Krivanek, O. L.; Zhou, W.; Chisholm, M. F.; Idrobo, J. C.; Lovejoy, T. C.; Ramasse, Q. M.; Dellby, N. Gentle STEM of Single Atoms: Low KeV Imaging and Analysis at Ultimate Detection Limits. In Low Voltage Electron Microscopy; John Wiley & Sons, Ltd: Chichester, UK, 2012; pp 119– 161
-
Uhlemann, S.; Müller, H.; Hartel, P.; Zach, J.; Haider, M. Thermal Magnetic Field Noise Limits Resolution in Transmission Electron Microscopy. Phys. Rev. Lett. 2013, 111, 046101 DOI: 10.1103/PhysRevLett.111.046101
-
Shao, Y.; Gan, Z.; Epifanovsky, E.; Gilbert, A. T. B.; Wormit, M.; Kussmann, J.; Lange, A. W.; Behn, A.; Deng, J.; Feng, X.; Ghosh, D.; Goldey, M.; Horn, P. R.; Jacobson, L. D.; Kaliman, I.; Khaliullin, R. Z.; Kuś, T.; Landau, A.; Liu, J.; Proynov, E. I.; Rhee, Y. M.M. Advances in Molecular Quantum Chemistry Contained in the Q-Chem 4 Program Package. Mol. Phys. 2015, 113, 184– 215, DOI: 10.1080/00268976.2014.952696
-
Mallikarjun Sharada, S.; Zimmerman, P. M.; Bell, A. T.; Head-Gordon, M. Automated Transition State Searches without Evaluating the Hessian. J. Chem. Theory Comput. 2012, 8, 5166– 5174, DOI: 10.1021/ct300659d
-
Sharada, S. M.; Bell, A. T.; Head-Gordon, M. A Finite Difference Davidson Procedure to Sidestep Full Ab Initio Hessian Calculation: Application to Characterization of Stationary Points and Transition State Searches. J. Chem. Phys. 2014, 140, 164115, DOI: 10.1063/1.4871660
-
Chai, J.-D.; Head-Gordon, M. Long-Range Corrected Hybrid Density Functionals with Damped Atom–Atom Dispersion Corrections. Phys. Chem. Chem. Phys. 2008, 10, 6615, DOI: 10.1039/b810189b